Abstract
Background: Patients (pts) with secondary acute myeloid leukemia (s-AML) have poor long-term outcomes following standard induction chemotherapy with 7+3. In 2017, a liposomal cytarabine and daunorubicin formulation (CPX-351) was FDA approved for upfront treatment of s-AML based on a pivotal phase 3 trial demonstrating improved overall survival in pts aged 60-75 years old (Lancet J et al; JCO 2018). Although CPX-351 treatment was subsequently approved for s-AML regardless of age, data in younger pts < 60 years who were not eligible for the pivotal trial is lacking. We sought to address this unanswered question by retrospective review of clinical experience since FDA approval at 6 large academic centers.
Methods: Medical records were retrospectively reviewed at Roswell Park Comprehensive Cancer Center, Memorial Sloan Kettering Cancer Center, Moffitt Cancer Center, Allegheny Health Network, University of Alabama Comprehensive Cancer Center, and Levine Cancer Institute to identify pts aged 18-59 years old with untreated s-AML defined as antecedent MDS or CMML, prior cytotoxic therapy, or AML with WHO defined myelodysplasia related changes (AML-MRC) treated with CPX-351 as induction therapy. Demographics, disease-specific variables, and outcomes were collected in accordance with the IRB approved protocol. Responses were defined per 2003 International Working Group (IWG) criteria. Demographic and baseline characteristics, treatment response, overall survival and adverse events were analyzed using descriptive statistics. Overall survival was estimated using Kaplan-Meier (KM) analysis.
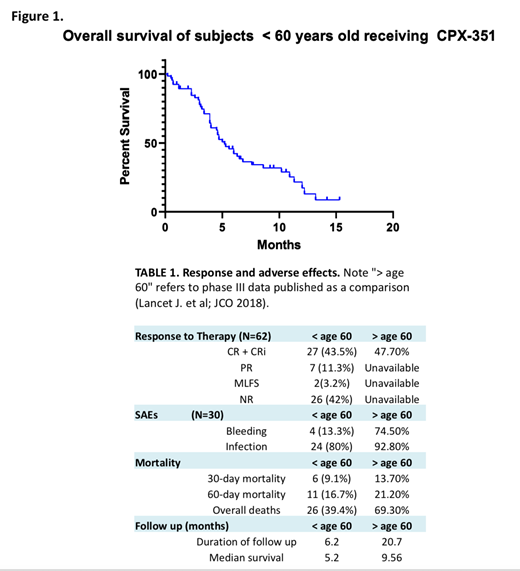
Results: Sixty-six pts with confirmed s-AML received CPX-351 therapy. Median age was 54.9 years (range 23 - 59), 37 were male (56%). The majority (65%, N=43) had AML-MRC, 23 (35%) had antecedent MDS, and 14 (21%) had treatment-related AML (t-AML). Sixteen pts had received prior hypomethylating therapy. Thirty pts had a complex karyotype (46%), and 10 patients were found to have a normal karyotype (15%). The most common molecular event was TP53 mutation observed in 19 pts (30%), followed by FLT3-ITD identified in 5 pts (8%). At the time of analysis, response assessments were available for 62 pts. Overall response rate (CR/CRi) was 43.5% with 19 CR (30.6%), 8 CRi (12.9%). Seven pts (11.3%) had PR, 2 pts (3.2%) with MLFS and the remainder (N=26, 42%) were non-responders (Table 1). Thirty-one pts have received an allogenic stem cell transplant. Of the 19 pts with TP53 mutation eligible for response assessment, 6 pts (31.5%) achieved CR, 2 pts (10.5%) had a PR, 1 pt (5.2%) had MLFS and the remaining 10 pts (52.6%) were primary refractory. In contrast, 21 (48.8%) of 43 TP53 wild-type pts achieved CR/CRi. The most common adverse event was infection (80%, 24/30) with 4 clinically significant bleeding events. Thirty-day mortality was 9.1%, with 60-day mortality of 16.7%. Overall survival in all evaluable pts (N=66) was 5.2 months (range 0.5 - 15.3 months) (Figure 1), with median follow up of 6.2 months.
Conclusions: This multi-institutional retrospective analysis demonstrates lower response rates (CR/CRi 43.5%) and shorter overall survival (5.2 mos) following CPX-351 therapy in young pts (<60) as compared to phase 3 results in older (60-75 years old) (Table 1). Potential explanations for this discrepancy include short follow up, small sample size, retrospective analysis, and high proportions of young pts with complex karyotype and TP53 mutant AML. Historically, patients < 65 years old with s-AML have had a reported overall survival of approximately 7 months. Further prospective investigation of CPX-351 vs 7+3 vs other approaches in younger pts with s-AML is clearly warranted.
Przespolewski: Jazz: Research Funding. Goldberg: Pfizer: Research Funding; Prelude Therapeutics: Research Funding; Aptose: Consultancy, Research Funding; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Aprea: Research Funding; Arog: Research Funding; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees; DAVA Oncology: Honoraria; Celularity: Research Funding; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees. Talati: Astellas: Speakers Bureau; Jazz: Speakers Bureau; Pfizer: Honoraria; BMS: Honoraria; AbbVie: Honoraria. Fazal: Takeda: Consultancy, Honoraria, Speakers Bureau; Stemline Therapeutics: Consultancy, Honoraria, Speakers Bureau; Taiho Pharmaceuticals: Consultancy, Honoraria, Speakers Bureau; Sanofi Genzyme: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria, Speakers Bureau; Karyopharm Pharmaceuticals: Consultancy, Honoraria, Speakers Bureau; Jazz Pharmaceuticals: Consultancy, Honoraria, Speakers Bureau; Janssen Oncology: Consultancy, Honoraria, Speakers Bureau; Incyte: Consultancy, Honoraria, Speakers Bureau; Glaxo Smith Kline: Consultancy, Honoraria, Speakers Bureau; Gilead Sciences: Consultancy, Honoraria, Speakers Bureau; Bristol Myers Squibb: Consultancy, Honoraria, Speakers Bureau; AMGEN: Consultancy, Honoraria, Speakers Bureau; Agios: Consultancy, Honoraria, Speakers Bureau. Vachhani: Incyte: Consultancy, Speakers Bureau; Pfizer: Consultancy; Blueprint Medicines: Consultancy; O'Neal Comprehensive Cancer Center, University of Alabama at Birmingham: Current Employment; Jazz Pharmaceuticals: Consultancy; Novartis: Consultancy; Abbvie: Consultancy; Seattle Genetics: Research Funding; Astellas Pharma: Speakers Bureau; CTI BioPharma Corp: Consultancy; Agios: Consultancy. Griffiths: Genentech: Research Funding; Boston Biomedical: Consultancy; Taiho Oncology: Consultancy, Honoraria; Novartis: Honoraria; Takeda Oncology: Consultancy, Honoraria; Astex Pharmaceuticals: Honoraria, Research Funding; Abbvie: Consultancy, Honoraria; Celgene/Bristol-Myers Squibb: Consultancy, Honoraria, Research Funding; Apellis Pharmaceuticals: Research Funding; Alexion Pharmaceuticals: Consultancy, Research Funding. Thompson: Novartis/ Bristol-Myers Squibb: Research Funding. Sweet: Gilead: Membership on an entity's Board of Directors or advisory committees; AROG: Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; Bristol Meyers Squibb: Honoraria, Membership on an entity's Board of Directors or advisory committees. Wang: Stemline Therapeutics: Consultancy, Honoraria, Other: Advisory board, Speakers Bureau; Pfizer: Consultancy, Honoraria, Other: Advisory Board, Speakers Bureau; Mana Therapeutics: Consultancy, Honoraria; Novartis: Consultancy, Honoraria, Other: Advisory Board; Kura Oncology: Consultancy, Honoraria, Other: Advisory board, steering committee, Speakers Bureau; Kite Pharmaceuticals: Consultancy, Honoraria, Other: Advisory Board; Jazz Pharmaceuticals: Consultancy, Honoraria, Other: Advisory Board; GlaxoSmithKline: Consultancy, Honoraria, Other: Advisory Board; Genentech: Membership on an entity's Board of Directors or advisory committees; BMS/Celgene: Membership on an entity's Board of Directors or advisory committees; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy, Honoraria, Other: Advisory board; DAVA Oncology: Consultancy, Speakers Bureau; Rafael Pharmaceuticals: Other: Data safety monitoring committee; Gilead: Consultancy, Honoraria, Other: Advisory board; Daiichi Sankyo: Consultancy, Honoraria, Other: Advisory board; PTC Therapeutics: Consultancy, Honoraria, Other: Advisory board; Genentech: Consultancy; MacroGenics: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal